Bringing a new drug to market has never been more challenging. On average, it takes 10 to 15 years and an estimated $1-2 billion to successfully move a novel therapeutic from discovery to approval. Despite unprecedented investments, most drug candidates never reach the finish line.
As development costs rise, timelines compress, and pipelines grow increasingly complex, pharmaceutical companies face mounting pressure to reduce risk earlier, and more strategically, across clinical development.
Incorporating patient genotyping into early stage clinical trials offers a powerful strategy for improving trial efficiency and increasing the probability of success. Once considered a “nice to have,” genetic insight is now a fundamental element of modern clinical trial design. Integrating genotyping into patient screening, stratification, and data analysis will improve clinical trial success by reducing safety issues, decreasing patient attrition, and generating highly-quality evidence to support regulatory submissions.
Why Clinical Trials Fail
Most investigational therapies that enter human studies will never reach approval. Success rates vary widely by therapeutic area and molecular modality. Overall, nearly 90% of clinical drug development programs fail, highlighting the need for new approaches to improve success rates. (1).
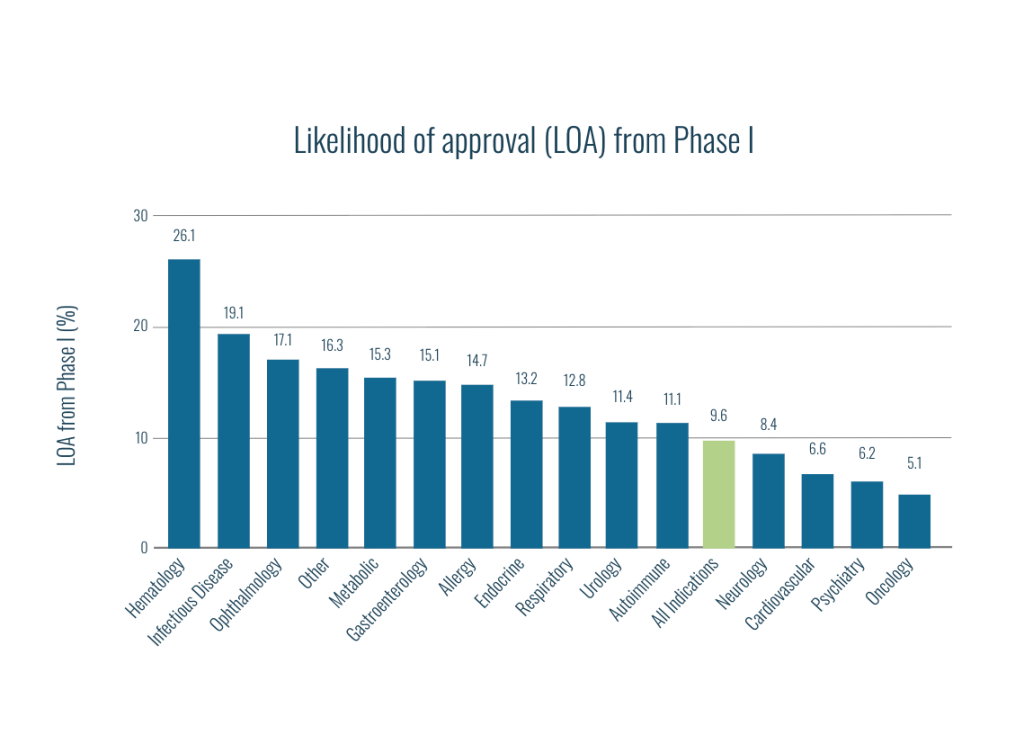
Approximately 53% of drugs fail in Phase I, most commonly due to safety, tolerability, or early efficacy concerns. Among those that advance to Phase II, only 28% overcome the so-called “Phase II hurdle,” where insufficient efficacy and persistent safety issues dominate failure rates. While success rates improve in Phase III, reaching roughly 55%, nearly half fall short of regulatory approval (2).
For every 20 drugs that enter Phase I trials, only one reaches the market. This highlights the critical importance of utilizing informed data-driven decision-making early in development, when changing course is still feasible and costs are comparatively contained.
The Root Causes of Clinical Trial Failure
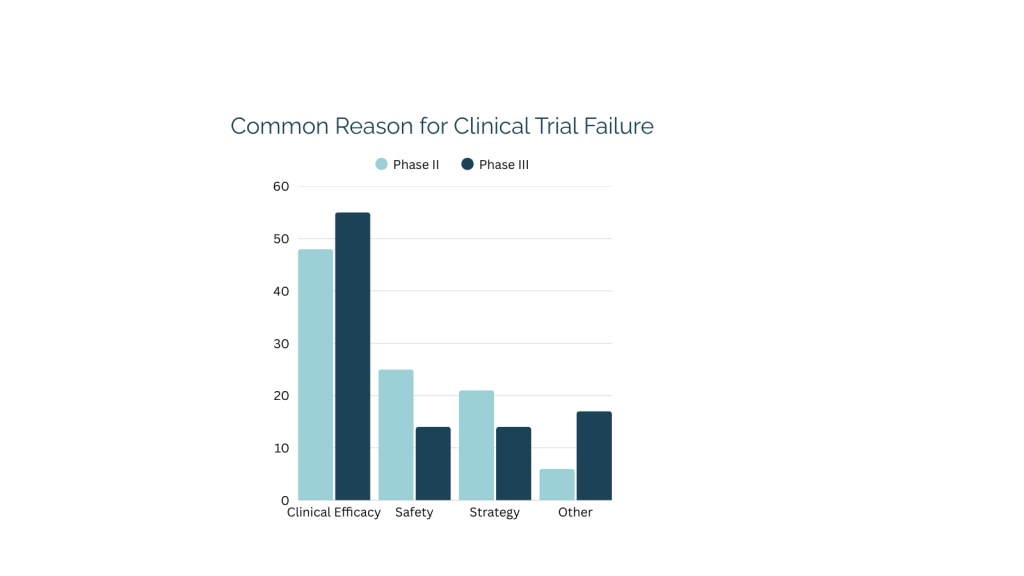
To improve clinical success rates, it is essential to understand why trials fail. A 2022 analysis published in Acta Pharmaceutica Sinica B identified four primary drivers of clinical trial failure, offering valuable insights into recurring failure patterns across the industry. Insufficient clinical efficacy emerged as the leading cause, accounting for approximately 40-50% of failures, followed by unmanageable toxicity at 30%. Additional contributors include poor drug-like properties (10-15%) and a lack of commercial viability or strategic misalignment (10%) (1).
Notably, the two most common drivers of failure, efficacy and safety, are also among the most biologically traceable. These are precisely the areas where genetic biomarker data can generate early, actionable insights. Genotyping is frequently incorporated into early stage clinical trials to better stratify patients, refine dosing strategies, and make more confident decisions before entering later-stage trials.
The Financial Cost of Getting it Wrong
The financial consequences of clinical trial failure are staggering. Estimates suggest that a single failed clinical program can cost between $800 million and $1.4 billion, excluding opportunity costs and pipeline delays (3). One of the most significant contributors is patient attrition.
Recruitment challenges are pervasive across the industry. Roughly 80% of clinical trials fail to meet their original enrollment timelines. For every 100 patients screened, only about seven complete a trial. Dropout rates can reach 30%, and replacing a single lost patient can cost nearly $20,000. This is compared to an average recruitment cost of $6,533 per enrollee (4).
Incorporating genotyping at the screening stage typically costs only a few hundred dollars per patient. Genotyping is a relatively small upfront investment that can prevent far more expensive downstream failures. This is particularly impactful for clinical trials facing avoidable safety issues or diluted efficacy signals. By identifying likely responders and excluding high-risk genotypes before randomization, trials leveraging genotyping benefit from reduced dropout rates, improved statistical power, and increased probability of success.
Genetic Biomarkers in Clinical Trials
Genetic biomarker analysis is increasingly used to address the root causes of trial failure, including safety concerns and lack of efficacy. In clinical research, biomarker strategies often include genotyping, gene expression analysis, and protein expression analysis. Each provides a distinct layer of biological insight.
Genotyping is used to identify DNA variants that influence drug-metabolizing enzymes, transporters, and receptors. In clinical trials, this information can help predict how a drug is absorbed, distributed, metabolized, and excreted (ADME). It can also be used to predict how patients are likely to respond to a medication, and which individuals may be at increased risk of adverse events. Technologies such as qPCR are commonly used to detect single-nucleotide polymorphisms (SNPs) that meaningfully impact drug response.
Gene expression analysis measures changes in the expression of target genes to validate mechanisms of action or target engagement, while protein expression analysis provides insight into downstream biological effects. These approaches often rely on tools such as RNA-sequencing, qPCR, ELISA, and Meso Scale Discovery (MSD). Together, these biomarker strategies enable sponsors to move beyond surface-level clinical endpoints and better understand why a therapy works, or fails, and for whom it is likely to be most effective.
CYP450 Genotyping: A Powerful Tool for Reducing Risk
The value of genotyping in clinical trials is perhaps most evident when evaluating cytochrome P450 (CYP450) enzymes. These enzymes are responsible for metabolizing the majority of drugs. Of the 57 known CYP450 enzymes, just six account for approximately 90% of drug metabolism: CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP3A4.
Genetic variation within these enzymes is both common and clinically significant. These genetic differences can dramatically alter drug exposure, efficacy, and toxicity, often explaining why a therapy performs inconsistently across patient populations.
By incorporating CYP450 genotyping into clinical trials, pharmaceutical companies can identify patients with altered metabolic capacity prior to enrollment, reducing the risk of adverse events, minimizing dropout, and generating actionable clinical data. When performed retrospectively, CYP450 genotyping helps explain unexpected outcomes and clarify safety or efficacy signals. Insights from retrospective genotyping can also strengthen regulatory strategy and future trial design.
A Real-World Example: CYP2C19 and Clopidogrel
The clinical relevance of CYP genotyping is well illustrated by clopidogrel, a widely prescribed antiplatelet therapy. As a prodrug, clopidogrel must be metabolized by CYP2C19 to become active. Patients carrying certain CYP2C19 variants are unable to efficiently convert the drug into its active form, resulting in diminished or absent therapeutic benefit.
In clinical trials, enrolling poor metabolizers alongside normal responders can obscure efficacy. It may also introduce avoidable safety concerns–outcomes that could be mitigated through upfront genetic screening.
When Genotyping is Overlooked: A Case Study in Delay and Cost
In one real-world case, a drug metabolized by CYP2C19 was initially denied FDA approval due to unresolved safety concerns. Throughout the clinical trial, patients reported adverse effects such as dizziness and lightheadedness, prompting regulators to question whether certain subpopulations were at elevated risk.
The FDA ultimately required CYP2C19 genotyping before reconsidering the application. Under a tight regulatory timeline, Genemarkers retrospectively genotyped approximately 1,800 patient samples across five clinical centers in just three months. The resulting data clarified the safety profile, supported resubmission, and led to FDA approval. Shortly thereafter, the company was acquired for $1 billion.
While a success story for this company, the program lost an estimated eight months, time that could have been avoided had genotyping been incorporated into the clinical studies earlier.
Bringing Genotyping into Clinical Trial Design
Genotyping can be implemented either prospectively or retrospectively depending on study goals and timelines. Prospective genotyping supports safer enrollment and helps identify patients who may require dose adjustments or alternative therapies. Retrospective genotyping allows sponsors to revisit stored samples to better understand unexpected outcomes without adding complexity upfront.
With high-throughput workflows capable of processing thousands of samples per week and noninvasive collection methods like buccal swabs, genotyping is both scalable and practical. At just 1-2% of the total cost of enrolling a patient, it represents a small investment with outsized potential to reduce clinical risk.
How Genemarkers Supports Genotyping for Clinical Trials
Genemarkers is a CLIA-certified, CAP-accredited, and GCLP-compliant contract research laboratory specializing in genomic and proteomic testing for drug discovery and development. We support clinical trials with high-quality, qPCR-based genotyping services designed to integrate seamlessly into existing workflows..
As regulatory expectations evolve and clinical development becomes increasingly data-driven, genetic insight is no longer optional, but now is a critical tool in modern clinical trial design.
Sources:
- Roth, C. (2019, May 9). The real cost of clinical trials. Praxis Communications. https://www.gopraxis.com/news/the-real-cost-of-clinical-trials/care benefits. Request a free consultation and quote to learn how you can incorporate biomarker research into your scalp care development.
- Sun, D., Gao, W., Hu, H., & Zhou, S. (2022). Why 90% of clinical drug development fails and how to improve it? Acta Pharmaceutica Sinica B, 12(7), 3049–3062. https://doi.org/10.1016/j.apsb.2022.02.002
- Why are clinical development success rates falling? (2024, May 16). Citeline. https://www.citeline.com/en/resources/why-are-clinical-development-success-rates-falling
- Roth, C. (2019, May 9). The real cost of clinical trials. Praxis Communications. https://www.gopraxis.com/news/the-real-cost-of-clinical-trials/
